Is Hi Polar Or Nonpolar
7.10.ane: Biology- Nonpolar Iodine and Polar Hydrogen Iodide
- Page ID
- 50812
Pure Covalent Bonds are those in which electrons are shared equally between the two atoms involved, as nosotros saw earlier, where the iodine molecule was given as an example:
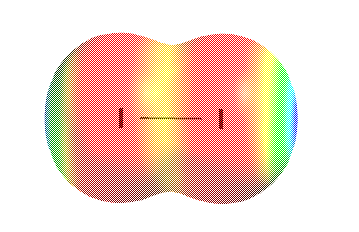
Electrostatic surface map for Iii
Information technology was also shown that replacing an I atom with a group I metallic decreased the covalent nature of the bond, while increasing its percent ionic character. The molecule is changed from a poisonous and bactericidal substance to salt-like white crystalline solids which may be more or less toxic depending on the particular metal chosen. IWC replica Réplica de reloj
Polar Covalent Compounds
But replacing one I atom in the majestic solid Iii with another nonmetal too makes a meaning difference. Replacing i of the iodine atoms with a hydrogen atom to make HI (hydrogen iodide) changes the chemistry significantly. Hullo is a colorless gas, and reacts with NaOH to give sodium iodide (used in iodized salt). Aqueous solution of Howdy are called hydroiodic acid, considering Hi dissolves extensively and readily in water to make acidic solutions by increasing the hydrogen ion (H+) concentration, while Iii is barely soluble in h2o. The polarity of the bond conspicuously has biological significance.
The Jmol model and electrostatic potential surfaces differ from those of I2 in several ways. Charge is no longer as distributed between the atoms; the I atom has an excess of well-nigh 0.05 electrons, on the average, over the number of electrons in the neutral atom, and so information technology has a charge of -0.05e. Even with electron shielding, the highly positive iodine nucleus pulls electrons toward itself more than the unmarried proton of the hydrogen nucleus attracts electrons. The H atom has lost 0.05 of an electron, then it has an electrostatic charge of +0.05 e. The molecule has two electrical "poles", and is called a dipole . The bail, in which electrons are non every bit shared, is called a polar covalent bond .
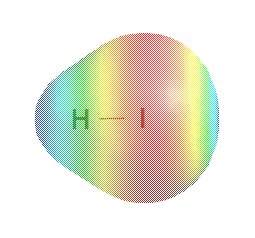
Electrostatic surface map for HI
In the H—I bond the I has an excess of 0.05 electrons and hence has a negative charge. This situation is frequently indicated as follows:
- δ+ δ-
- H----I δ=0.05
or Hδ + Iδ -
The Greek alphabetic character δ (delta) is used here to indicate that electron transfer is not complete and that some sharing takes place. If the transfer had been consummate, δ would accept been 1.0. Considering the Li—H bond is only partially negative at the one stop and partially positive at the other, nosotros often say that the bond is polar or polar covalent, rather than 100 percent ionic.
Elements in the upper right of the periodic table, which are pocket-sized because the large nucleus contracts the valence shell, grade much more than polar bonds with H. For example, HF has a δ value of 0.43, compared with δ = 0.05 for HI.
- δ+ δ-
- H----F δ=0.43
Lets see how to calculate the δ value for Hello. The data for this example could be obtained from the Jmol model in a higher place, only there are a number of different methods for calculating charges, and the one used here may not exist appropriate for this calculation. Even so, the bond altitude in Howdy can be measured past right clicking one the Jmol model, choosing "measurements", so clicking on the atoms in sequence. We'll use empirically (from experiment) measured values here:
Instance 1 The dipole moment of the HI molecule is institute to be 1.34 × 10–30 C chiliad, while the H―I distance is 165.0 pm. Find the partial charge on the H and F atoms.
Solution Rearranging Eq. (1) from Polarizability, we take
-
- \(Q=\frac{\mu }{r}\)
Thus the apparent charge on each end of the molecule is given past
-
- \(Q=\frac{\text{1}\text{.34 }\times \text{ 10}^{-\text{30}}\text{ C chiliad}}{\text{165}\text{.0 }\times \text{ x}^{-\text{12}}\text{ m}}=\text{8}\text{.x }\times \text{ 10}^{-\text{21}}\text{ C}\)
Since the charge on a single electron is 1.6021 × 10-xix C, we take
-
- \(\delta =\frac{\text{8}\text{.10 }\times \text{ 10}^{-\text{21}}}{\text{one}\text{.6021 }\times \text{ 10}^{-\text{19}}}=\text{0}\text{.051}\)
Then δ = 0.051.
Information technology is worth noting in the above example that the dipole moment measures the electric imbalance of the whole molecule and not just that of the H―I bonding pair. In the HI molecule there are 4 valence electron pairs, with the three lone pairs on the right of the I atom as well contributing to the overall negative charge of I.
Is Hi Polar Or Nonpolar,
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Book%3A_ChemPRIME_(Moore_et_al.)/07%3A_Further_Aspects_of_Covalent_Bonding/7.10%3A_Polar_Covalent_Bonds/7.10.01%3A_Biology-_Nonpolar_Iodine_and_Polar_Hydrogen_Iodide
Posted by: lieberarequire.blogspot.com



0 Response to "Is Hi Polar Or Nonpolar"
Post a Comment